Atomic emission spectrum worksheet blank12/9/2023 
We have different line spectrum that can be formed now with a line emission spectrum. So click on the next video and let's take a look at the different types of a mission line spectrum. So with these emission lines will learn about what their names are and between what shells does the electron travel. Spectra is examined as a series of atomic emissions and realize that an atomic emission involves releasing energy as an electron goes from a higher shell number to a lower shell number. Now, with Lina Message Coalition Spectra, we have each line of an emission. Now the prison itself, it transforms wavelengths into discrete lines on an emission spectrum. So the slit here is focusing the light and allowing us to separate it into these different bands of color. Now we're going to say the slate is just a long, narrow cut used to spread closely packed wavelength, which can later be determined. This emission spectrum can be used to determine what shells has the electron traveled through. The slit itself focuses that light and has it passed through a prism? Then that prism basically creates different colored lines on which we call in a mission spectrum. 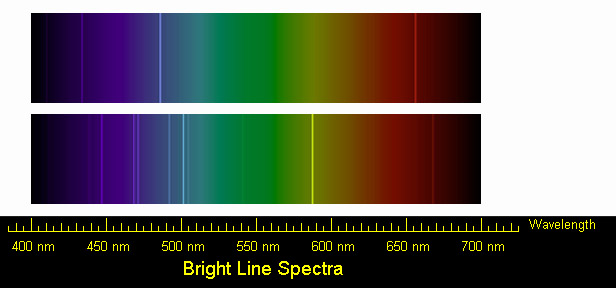
So doing this transition would emit energy, and that energy could be emitted as light. And with a mission, we have the releasing of energy. So we're going from a higher shell number. Now here we have our electron going from Ah, higher shell back down to our shell number one. Here we have shell one shell to and then here we have infinite number of possible shelves for any given. We have the nucleus with its protons and it's neutrons, and we have our different shelves. Now, here we have the representation of an atom in the middle. 
Emission Spectra is a Siris of lines formed when omitted light is focused by a slit and passes through a prism.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
